About InnovaBurn®
About InnovaBurn®
InnovaBurn® was cleared by the FDA on September 29, 2022, via the 510(k) pathway – K213716.
InnovaBurn® is an important advancement in the treatment of partial-thickness, second-degree burns, giving physicians a placental-derived medical device that offers some of the largest sizes in the industry. InnovaBurn® requires no preparation, no specific placement orientation, no tissue tracking and no special storage.
Contact Us
Sign up today to get InnovaBurn® to treat your patients.
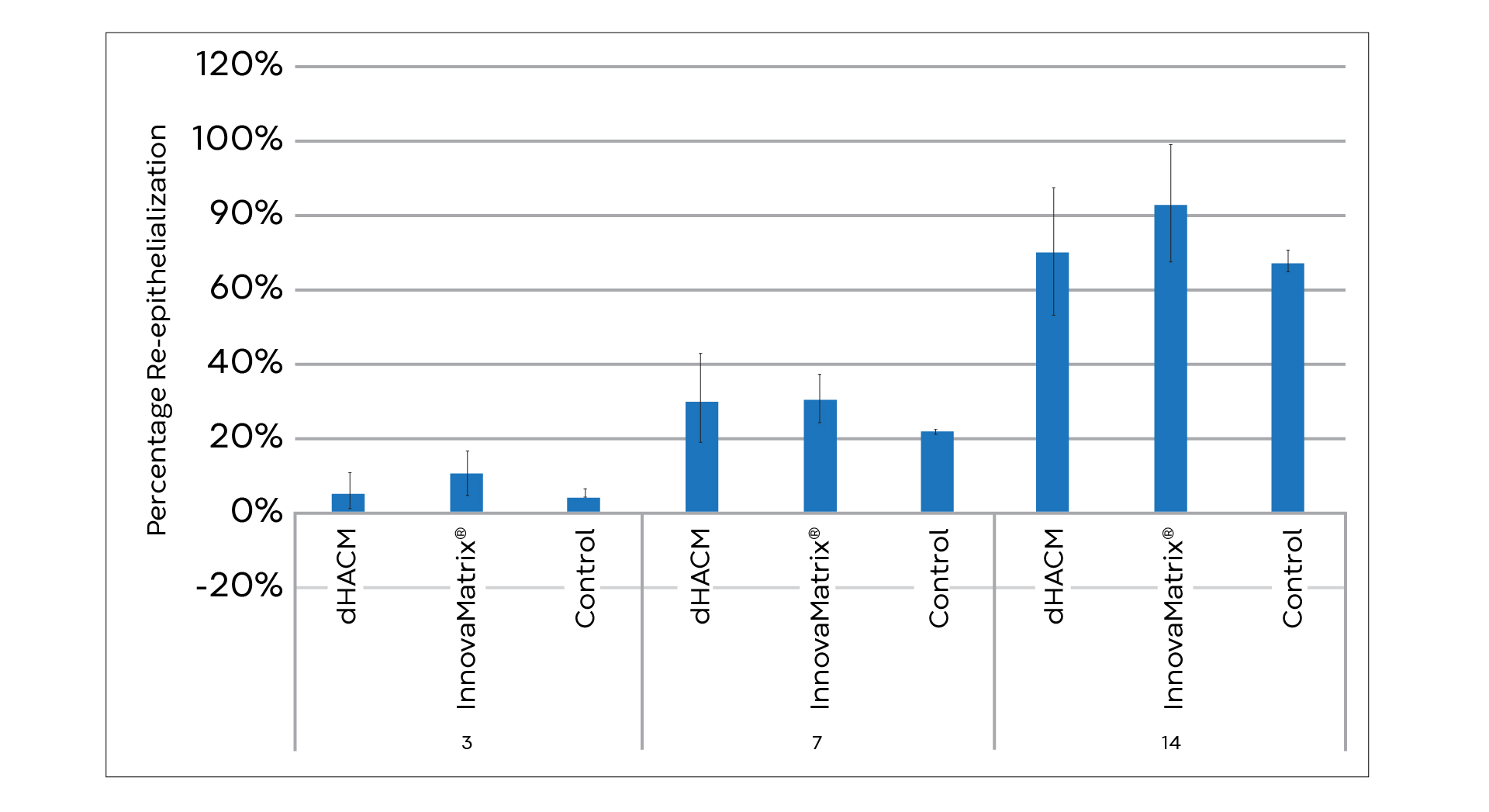
InnovaBurn® brings advanced ECM technology to patients with partial-thickness, second-degree burns. As a next-generation technology, InnovaBurn® offers the inherent benefits of the placenta1,2 plus the quality control, reliability, and safety profile of a medical device.3,4 This is good news for the thousands of patients who sustain burn injuries each year. Manufactured with our proprietary TriCleanse™ Placental Extracellular Matrix (ECM) Process, InnovaBurn® is state-of-the-art technology for management of hard-to-heal wounds, including partial-thickness, second-degree burns, surgical wounds, and traumatic wounds.

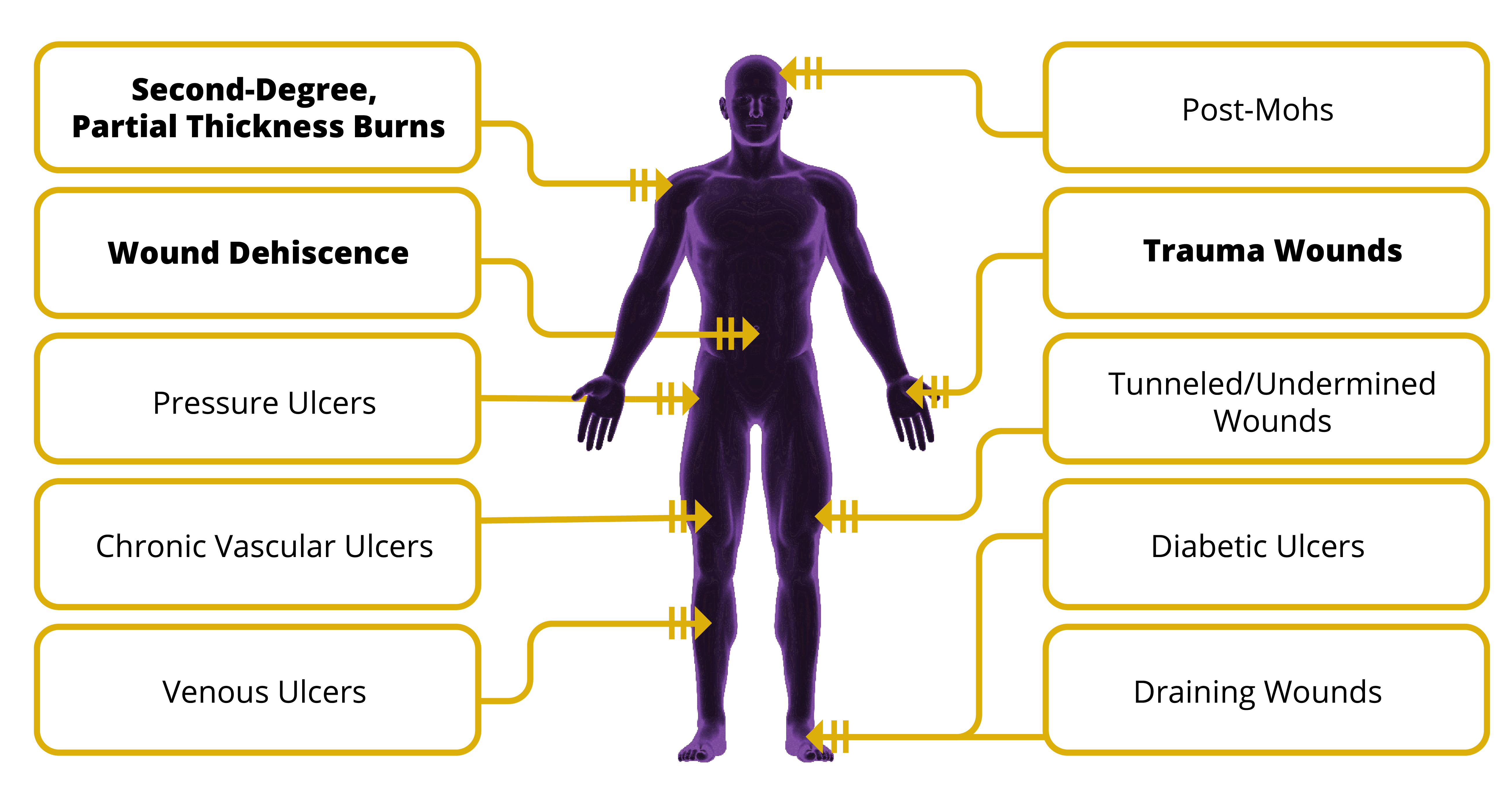
Indicated For the Management of Wounds Including:*
*See package insert for full list of indications.
Material Selection
While the benefits of placental-derived allografts are well documented,1,2 human-derived grafts can be variable due to the unique medical histories and social behaviors of each donor.5-12 Knowing this challenge, a porcine source was selected because it can be controlled for age, diet, health, and activity level, thereby reducing the variability of the raw material used to manufacture InnovaBurn® and other InnovaMatrix® products.
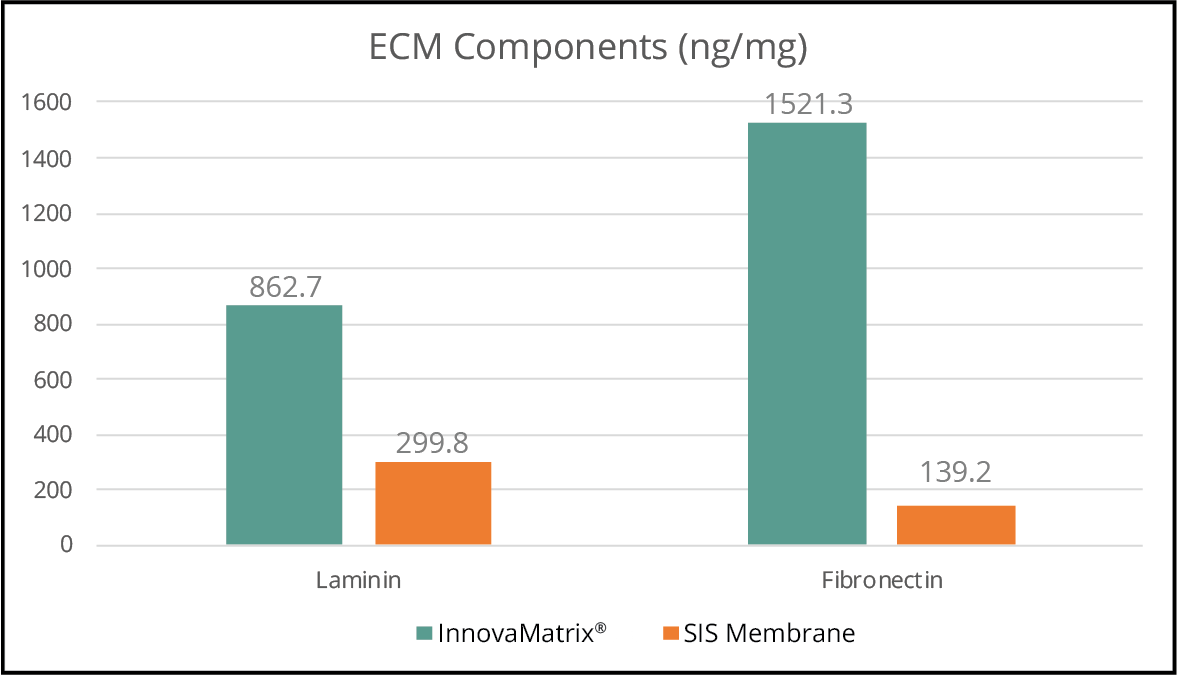
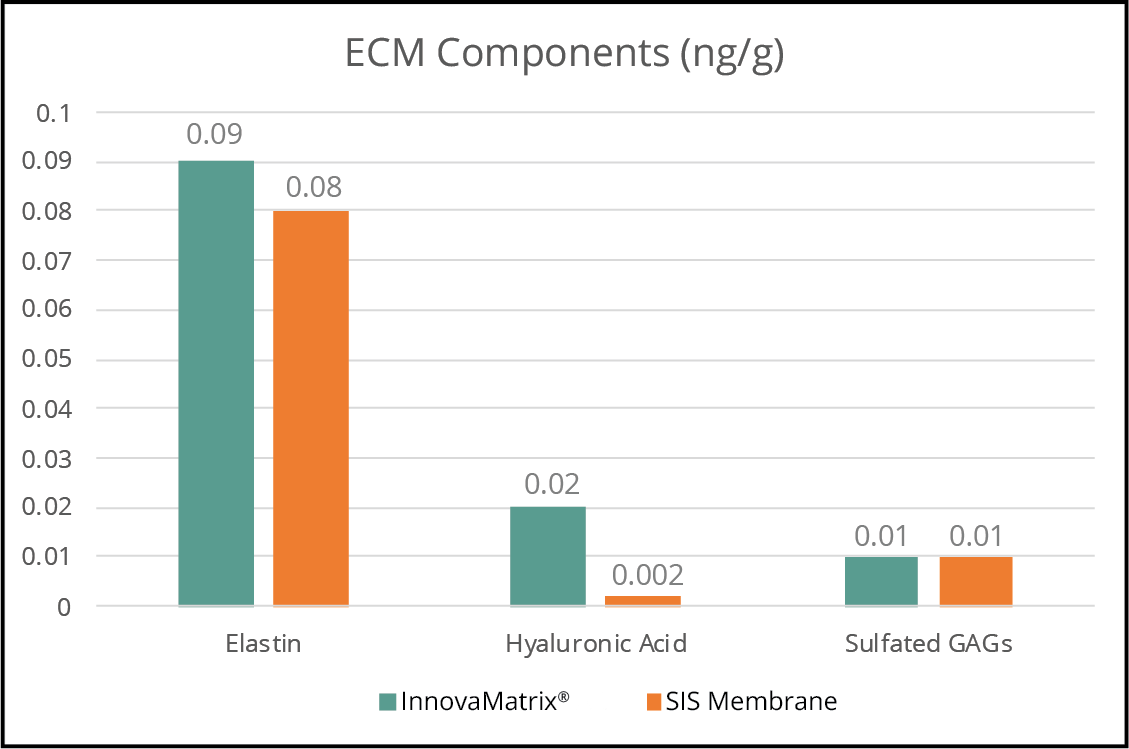
Our extracellular matrix medical devices use a unique placental source material with a high content of collagen, hyaluronic acid, and naturally embedded functional proteins.3,4
Balancing Decellularization Efficiency and the Denaturing of ECM Proteins
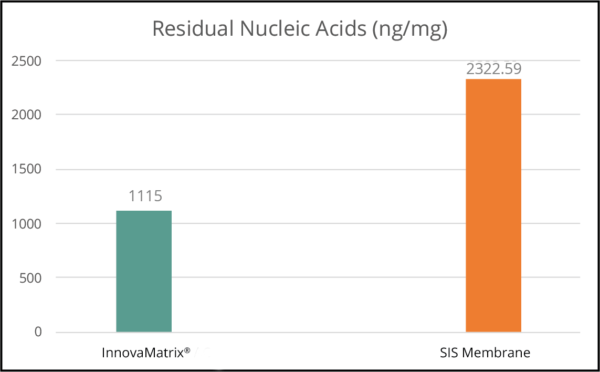
As a placental medical device processed with the proprietary TriCleanse™ Process, InnovaMatrix® was extensively biochemically characterized to identify and quantify the different structural and functional proteins. These results were then compared to an SIS membrane device as a base line for comparison.
Cellular Debris Comparison4
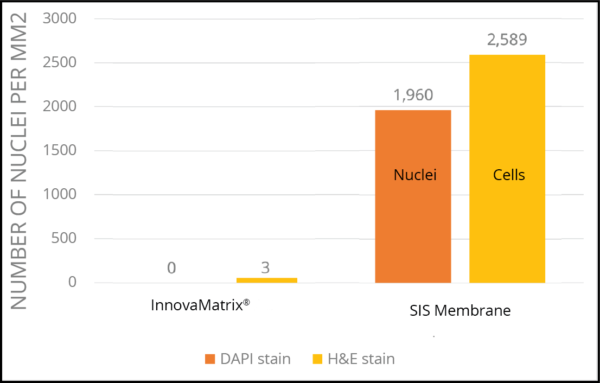
The presence of intact cells and nuclei creates an immunogenic response as cellular antigens are recognized as foreign by the host’s immune system.
The immune system has the innate capability to recognize nucleic acids not contained within the nucleus via pattern recognition receptors and mount an inflammatory response to their presence.
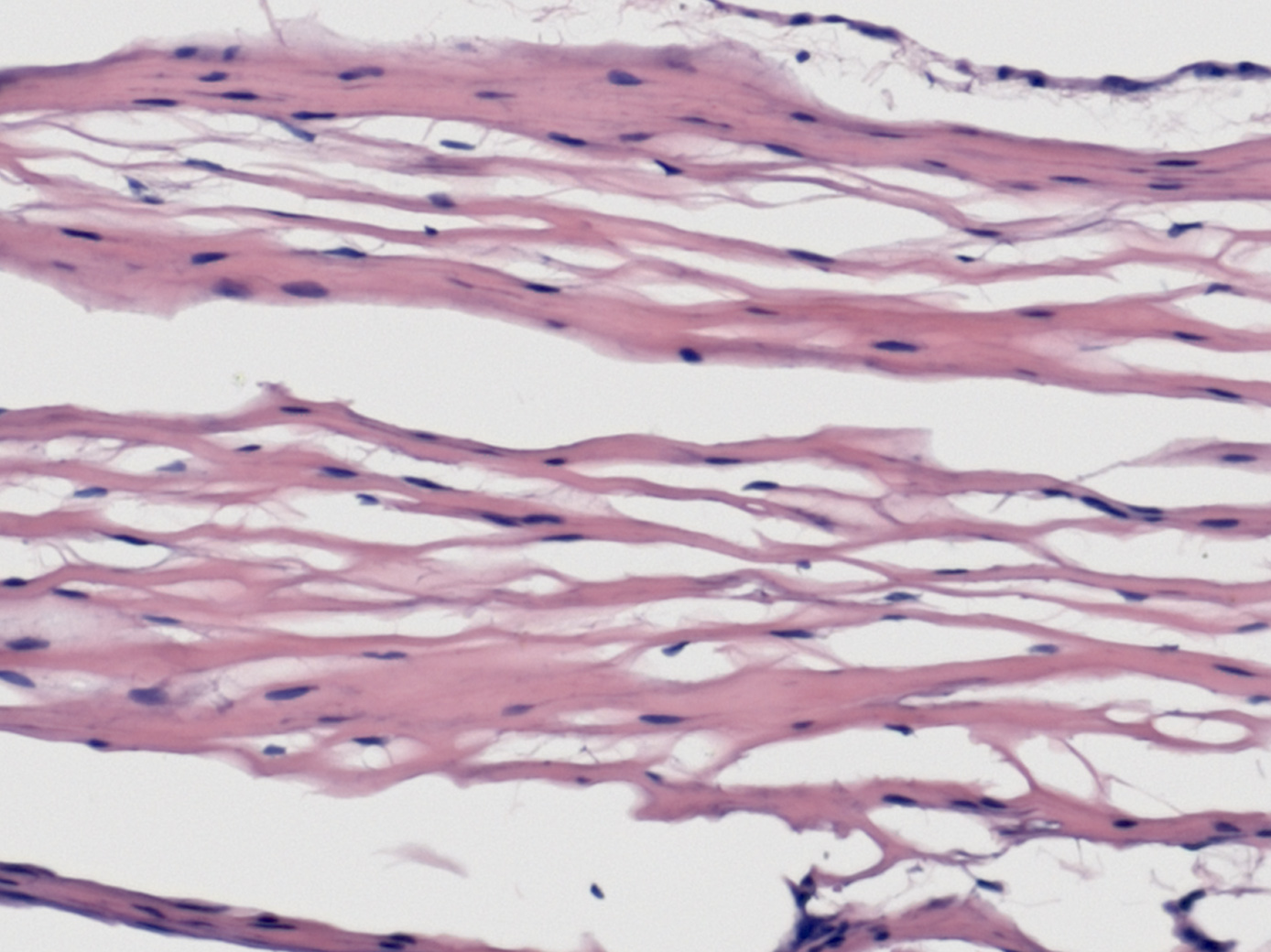
Hematoxylin and Eosin Staining4
Hematoxylin and Eosin (H&E) staining is commonly used to stain tissue for histology evaluation.17 H&E stains nuclei blue and extracellular matrix and cytoplasm pink and other tissues shades in between.
InnovaMatrix®

The H&E staining shows a minute quantity of residual nuclei in the finished product.
SIS Commercial Membrane
The H&E staining shows clearly stained blue nuclei are distributed throughout the finished product.
HCT/P Commercial Graft
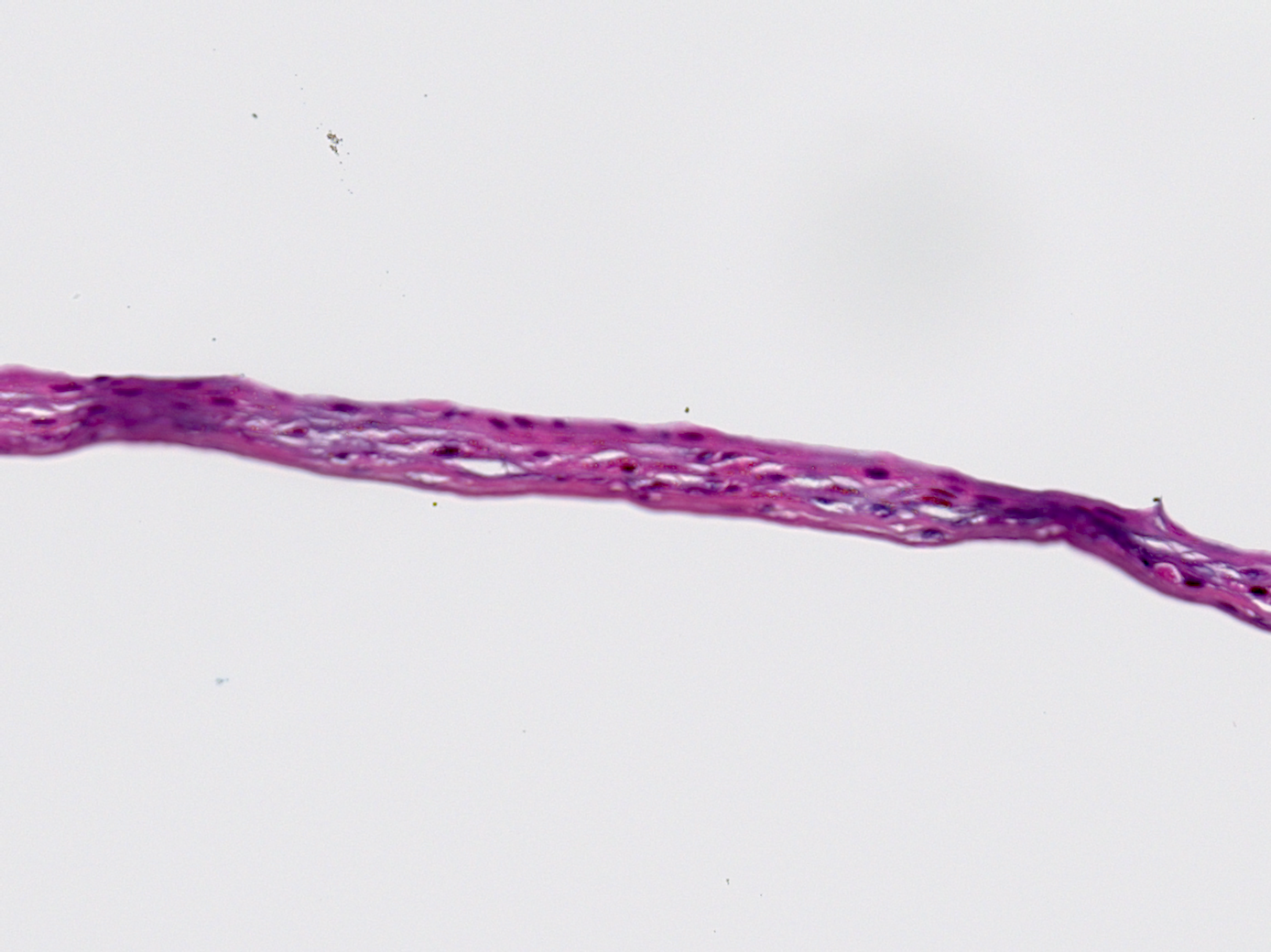
The H&E staining shows blue stained nuclei present in the entirety of the graft with areas of high concentration.

Indicated for the Management of Wounds Including:
*See package insert for full list of indications.
- Partial and full-thickness wounds
- Pressure ulcers
- Venous ulcers
- Diabetic ulcers
- Chronic vascular ulcers
- Tunneled/undermined wounds
- Surgical wounds (donor sites/ grafts, post-Mohs surgery, post-laser surgery, podiatric, wound dehiscence)
- Trauma wounds (abrasions, lacerations, and skin tears)
- Partial-thickness second degree burns
- Draining wounds
The device is intended for one-time use.
Contraindications
This device is derived from porcine collagen and should not be used on patients with sensitivity or allergy to porcine materials; sensitivity or allergy to collagen; or active or latent infection in or around the application site.
This device is not indicated for use in third degree burns.
Download our
InnovaBurn® Documents
Use the arrows to navigate.
MKT-2023-0051-22 V01
- Shaifur Ra, M., Islam, R., Asaduzzama, S.M., & Shahedur R, M. (2015). Properties and Therapeutic Potential of Human Amniotic Membrane. Asian Journal of Dermatology, 7(1), 1-12. doi: 10.3923/ajd.2015.1.12
- Fairbairn, N. G., Randolph, M. A., & Redmond, R. W. (2014). The clinical applications of human amnion in plastic surgery. Journal of Plastic, Reconstructive & Aesthetic Surgery, 67(5), 662-675.
- K193552 510(k) Summary
- Data on file -RDR-002
- Cardinal, L. J. (2015). Central tendency and variability in biological systems. J Community Hosp Intern Med Perspect, 5(3), 27930. doi:10.3402/jchimp.v5.27930
- Collier, A. C., Tingle, M. D., Paxton, J. W., Mitchell, M. D., & Keelan, J. A. (2002). Metabolizing enzyme localization and activities in the first trimester human placenta: the effect of maternal and gestational age, smoking and alcohol consumption. Hum Reprod, 17(10), 2564-2572. doi:10.1093/humrep/17.10.2564
- DuBois, B. N., O’Tierney-Ginn, P., Pearson, J., Friedman, J. E., Thornburg, K., & Cherala, G. (2012). Maternal obesity alters feto-placental cytochrome P4501A1 activity. Placenta, 33(12), 1045-1051. doi:10.1016/j.placenta.2012.09.008
- Huuskonen, P., Amezaga, M. R., Bellingham, M., Jones, L. H., Storvik, M., Hakkinen, M., . . . Pasanen, M. (2016). The human placental proteome is affected by maternal smoking. Reprod Toxicol, 63, 22-31. doi:10.1016/j.reprotox.2016.05.009
- McRobie, D. J., Glover, D. D., & Tracy, T. S. (1998). Effects of gestational and overt diabetes on human placental cytochromes P450 and glutathione S-transferase. Drug Metab Dispos, 26(4), 367-371. Retrieved from https://www.ncbi.nlm. nih.gov/pubmed/9531526
- O’Huallachain, M., Karczewski, K. J., Weissman, S. M., Urban, A. E., & Snyder, M. P. (2012). Extensive genetic variation in somatic human tissues. Proc Natl Acad Sci U S A, 109(44), 18018-18023. doi:10.1073/pnas.1213736109
- Paakki, P., Stockmann, H., Kantola, M., Wagner, P., Lauper, U., Huch, R., . . . Pasanen, M. (2000). Maternal drug abuse and human term placental xenobiotic and steroid metabolizing enzymes in vitro. Environ Health Perspect, 108(2), 141-145. doi:10.1289/ehp.00108141
- Strolin-Benedetti, M., Brogin, G., Bani, M., Oesch, F., & Hengstler, J. G. (1999). Association of cytochrome P450 induction with oxidative stress in vivo as evidenced by 3-hydroxylation of salicylate. Xenobiotica, 29(11), 1171-1180. doi:10.1080/004982599238038
- Malinda KM, Wysocki AB, Koblinski JE, Kleinman HK, Ponce ML. Angiogenic laminin-derived peptides stimulate wound healing. Int J Biochem Cell Biol. 2008;40(12):2771-80;
- Grinnell F, Billingham RE, Burgess L. Distribution of fibronectin during wound healing in vivo. J Invest Dermatol. 1981 Mar;76(3):181-9
- Litwiniuk M, Krejner A, Speyrer MS, Gauto AR, Grzela T. Hyaluronic Acid in Inflammation and Tissue Regeneration. Wounds. 2016 Mar;28(3):78-88
- Final Report: Evaluation of the host response to wound care products in a chronic wound model
- Hinton, J. P., Dvorak, K., Roberts, E., French, W. J., Grubbs, J. C., Cress, A. E., . . . Nagle, R. B. (2019). A Method to Reuse Archived H&E Stained Histology Slides for a Multiplex Protein Biomarker Analysis. Methods Protoc, 2(4). doi:10.3390/mps2040086